Evaluating Feed Components and Finished Feeds: Part 3
Tim Herrman
Extension State Leader
Grain Science and Industry
Kansas State University
Laboratories
When selecting a laboratory, price should not be the only consideration. It is important to find out which professional association laboratory personnel belong to and analytical techniques used. Official methods are tested and approved by members of these professional organizations: i.e., Association of Official Analytical Chemists (AOAC) or American Association of Cereal Chemists (AACC).
Some membership affiliations to look for include: AOAC, AACC, American Chemical Society, American Oil Chemists Society, National Oilseed Processors Association, American Fats and Oils Association, National Institute of Oilseed Products, and NFIA. Also check to see if the lab participates in check sample programs provided by the Association of American Feed Control Officials (AAFCO), American Feed Ingredients Association, and other professional organizations.
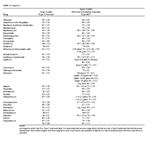
 Table 2 presents a partial list of labs and mailing address, phone number, and web address where ser- vices and prices are listed. Laboratories and services appearing in this publication are used for identification only. No endorsement is intended, nor is criticism implied of laboratories not mentioned. Most labs will analyze for individual components as well as offer special rates for grouped analyses. One such common group analysis is proximate analysis. Proximate analysis consists of moisture, crude protein, crude fat, crude fiber, ash, and nitrogen-free extract.
Table 2 presents a partial list of labs and mailing address, phone number, and web address where ser- vices and prices are listed. Laboratories and services appearing in this publication are used for identification only. No endorsement is intended, nor is criticism implied of laboratories not mentioned. Most labs will analyze for individual components as well as offer special rates for grouped analyses. One such common group analysis is proximate analysis. Proximate analysis consists of moisture, crude protein, crude fat, crude fiber, ash, and nitrogen-free extract.
Analytical Variation (AV)
AAFCO has established analytical variation (AV) guidelines in order to assist control officials in making decisions regarding marginally acceptable products (AAFCO, 2000). These variances are intended to allow for inherit variability in sampling and laboratory analyses. They are not intended to allow for deficiencies or excesses in a product or poor analytical techniques.
 Table 3 shows the analytical variances for some of the common ingredients. If the assay indicates that the ingredient is outside the analytical variance, the feed does not conform to label requirements. The concentration range indicates for what inclusion rate (level) the Analytical Variation Percentages (AV%) apply; e.g., moisture AV% applies to feed containing between 3 and 40 percent moisture. The AV% can be calculated using the following steps:
Table 3 shows the analytical variances for some of the common ingredients. If the assay indicates that the ingredient is outside the analytical variance, the feed does not conform to label requirements. The concentration range indicates for what inclusion rate (level) the Analytical Variation Percentages (AV%) apply; e.g., moisture AV% applies to feed containing between 3 and 40 percent moisture. The AV% can be calculated using the following steps:
Step 1: Multiply the expected or guaranteed value by the value derived from the formula in Table 3 in the AV% column. Convert the AV% value to the decimal equivalent (move the decimal two places to the right).
Step 2: Add and subtract the value obtained in Step 1 to the expected or guaranteed value.
Example:
Suppose a sample of soybean meal was submitted for protein analysis. If the expected or guaranteed protein content was 44%, the acceptable range would be 42.9-45.1.
Step 1:
44 x {(20 / 44 + 2) / 100} = 1.08
Step 2:
44 - 1.08 = 42.9
44 + 1.08 = 45.1
Drug Analysis
The FDA's Current Good Manufacturing Practices (CGMPs) stipulate that periodic assays of medicated feeds for drug components shall be performed as a means of monitoring the manufacturing process. Each category II Type A drug must be sampled and assayed three times each year. For medications containing a combination of drugs, perform the assay on only one drug each time and rotate the drugs analyzed. If the results of these assays are outside the permissible limits listed in Table 4 and Table 5 (Feed Additive Compendium, 2001), an investigation and corrective action must be implemented.
The CGMPs also stipulate that "corrective action shall include provisions for discontinuing distribution where the medicated feed fails to meet the labeled drug potency. Distribution and subsequent production of the particular feed shall not begin until it has been determined that proper control procedures have been established."
Many commercial feed mills and on-farm feed processors are not required by law to conduct drug assays since they are not registered (for further information, refer to Kansas State University Extension bulletins MF-2042 and MF-2043 by Herrman et al., 2000). However, routine inspection of finished feed for drug potency is a good business practice.
Unfortunately, the CGMPs do not provide advice on how to investigate high or low drug potency. Typical sources (or reasons) for out-of-tolerance assays include the following:
- the medicated article has lost its drug potency,
- incorrect weighing of the medicated article,
- poor mixing of the medicated article into the feed,
- poor sampling technique.
One way to perform drug assays on medicated feed is to use the same sampling technique used to conduct mixer performance tests. A complete description of this procedure is listed in the bulletin titled Testing Mixer Performance (Herrman and Behnke, 2001). The procedure involves collecting samples from 10 representative locations in the mixer. Combine the samples to form a single composite sample for the drug assay. One half of the composite sample should be retained for a minimum of three months, in the event the first sample is out of tolerance. Also, collect and retain a sample of the medicated article. If the feed is out of tolerance, submit a sample of the medicated article and the retained portion of the feed sample for drug analysis. Note: similar procedures may be followed for identifying the source of variation of other ingredients in complete feed.
The CGMPs also specify methods to avoid cross- contamination of feed when using medicated articles to produce medicated feed. These methods include flushing, sequencing, and equipment clean-out. Procedures to avoid drug cross-contamination are discussed in detail in MF-2055.
Utilizing Assay Results
After investing considerable time and capital to collect a representative sample and have it analyzed, the feed processor must manage the information.
Correct information management will assist in:
- detecting ingredient/product variation,
- evaluating suppliers,
- determining the discount for substandard product,
- fine-tuning feed rations,
- explaining animal performance problems,
- meeting FDA CGMPs (if feed mill is licensed).
A simple way to utilize information involves record- ing lab results in table form (either by hand or on a computer spreadsheet program). Columns in the table should include the date material was received, lab number assigned to the sample, ingredient supplier, and assay results (e.g., protein, moisture). Separate data sheets should be kept for each ingredient type (e.g., grain, protein, drug). These results should be regularly compared with contract specifications to ensure suppliers are shipping ingredients that meet or exceed quality criteria. Summarize data by month and supplier to detect noticeable trends.
The use of Statistical Process Control (SPC) to evaluate assay data provides an additional management tool from which to control variability in finished feed, thus improving product quality and profitability. For further information, read the bulletin titled Statistical Process Control: Techniques for Feed Manufacturing (Herrman, 2001).
Summary
Feed ingredients should be routinely evaluated to ensure they are safe, they contain the correct amount of the specified nutrient, and to ensure the finished feed quality will optimize animal performance. A list of ingredients, their important nutritional components, where they can be tested, and how to interpret this information is provided in this bulletin. Permitted analytical variation (PAV) guidelines are included to explain how to identify deficiencies or excesses of an ingredient in a product. If the assay indicates the ingredient is outside the PAV, the feed does not conform to label requirements. Techniques for identifying the source of variation and corrective actions are discussed.
Other Parts to this Article